The standard heat of formation standard enthalpy of formation of a compound is defined as the enthalpy change for the reaction in which elements in their standard states produce products. DeltaH_diss -q_solution The minus sign is used here because heat lost carries a negative sign.

Week 23 C Pearson Education Ltd 2009 This Document May Have Been Altered From The Original Use The Enthalpy Change Of Solution Of A Simple Ionic Solid Ppt Download
Energy Changes In Solutions

12 Thermodynamics 12 1 Types Of Enthalpy Change 12 2 Born Haber Cycles 12 3 Enthalpy Changes Enthalpy Of Solution 12 4 Mean Bond Enthalpy 12 5 Entropy Ppt Download
Specific enthalpy enthalpy per kg of dry air of moist air is the sum of the specific enthalpy of dry air and the specific enthalpy of the water vapor in the air.
Enthalpy change of solution. Qrxn where n is the number of moles of a specific reactant or product and ΔH is the enthalpy change of the reaction in kJmol. Others dissolve exothermically for example NaOH. For very weak acids like hydrogen cyanide solution the enthalpy change of neutralisation may be much less.
Enthalpy is an extensive quantity so the amount of heat generated by the reaction is given by the expression. The temperature of the water rose to 275C. The change in enthalpy is directly proportional to the number of reactants and products so you work this type of problem using the change in enthalpy for the reaction or by calculating it from the heats of formation of the reactants and products and then multiplying this value times the actual quantity in moles of material that is present.
Using q m x cp x T calculate energy change for quantities used 2. The unit of enthalpy change is Kilojoule per mole KJ mol-1. Enthalpy Review You may wish to review the Laws of Thermochemistry and Endothermic and Exothermic Reactions before you begin.
1 g cm-3 Solution. Enthalpy of bond dissociation is defined as the enthalpy change when one mole of covalent bonds of a gaseous covalent compound is broken to form products in the gaseous phase. The standard enthalpy change of neutralization is the enthalpy change when solutions of an acid and an alkali react together under standard conditions to produce 1 mole of water.
Gibbs combined enthalpy change and entropy change by the following equation ΔG ΔH - TΔS ΔG is change in the free energy ΔH is the change in enthalpy ΔS is the change in entropy and T. In an experiment 12 g of sodium hydroxide pellets NaOH s were dissolved in 100 mL of water at 25C. Enthalpies of solution may be either positive or negative - in other words some ionic substances dissolved endothermically for example NaCl.
Enthalpy of solution or heat of solution is expressed in kJmol and it is the amount of heat energy that is released or absorbed when a solution is formed. Since the energy of the universe is conserved the total. At 25C and 1 atm 1013 kPa the standard state of any element is solid with the following exceptions.
Thus the series of steps must have individual enthalpy changes that add up to the total change in enthalpy for the process. More specifically you can assume that. For example one source which gives the enthalpy change of neutralisation of sodium hydroxide solution with HCl as -579 kJ mol-1 gives a value of -561 kJ mol-1 for sodium hydroxide solution being neutralised by ethanoic acid.
To find the heat absorbed by the solution you can use the equation. Calculating the enthalpy change of reaction Hr from experimental data General method 1. The heat of neutralisation between sulphuric acid and potassium hydroxide solution is -546 kJ mol-1.
There are three steps in solvation. 42 J g-1 C-1. Enthalpy of Solution Heat of Solution Example.
3 m is the mass mass of the reactants mass of water mass of calorimeter C is the. The enthalpy of solution is most often expressed in kJmol at constant temperature. The change in enthalpy of a reaction ΔH is the same if the reaction happens in a single step or in a series of steps.
There are two types of enthalpy changes exothermic negative enthalpy change and endothermic positive enthalpy change. The solution including the reactants and the products and the calorimeter itself do not undergo a physical or chemical change so we need to use the expression for specific heat capacity to relate their change in temperature to the amount of heat q cal that they have exchanged Eqn. The relationship between the change in the internal energy of the system during a chemical reaction and the enthalpy of reaction can be summarized as follows.
The energy change can be regarded as being made of three parts the endothermic breaking of bonds within. Calculate the enthalpy change heat of solution for the reaction in kJ mol-1 of solute. Specific heat capacity of solution.
The enthalpy of the products assuming that the reaction goes to completion and the initial enthalpy of the system namely the reactants. This enthalpy change example problem is the enthalpy change as ice changes state from solid to liquid water and finally to water vapor. Dissolving calcium chloride and sometimes endothermic eg.
H ha Hhg. Sometimes this is exothermic eg. It is the difference between the enthalpy after the process has completed ie.
Enthalpies of solution may be either positive or negative - in other words some ionic substances dissolved endothermically for example NaCl. Where ha is the specific enthalpy of dry air H is the. The enthalpy of solution enthalpy of dissolution or heat of solution is the enthalpy change associated with the dissolution of a substance in a solvent at constant pressure resulting in infinite dilution.
Notice that enthalpy change of neutralization is always measured per mole of water formed. This is given by the equation. Work out the moles of the reactants used 3.
Others dissolve exothermically for example NaOH. The heat given off or absorbed when a reaction is run at constant volume is equal to the change in the internal energy of the system. Enthalpy change of solution and Enthalpy change of Hydration When ionic compounds dissolve in water there is usually a temperature change.
A student carried out an experiment to investigate the change in temperature during a titration between sodium hydroxide solution and hydrochloric. The enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution. Divide q by the number of moles of the reactant not in excess to give H 4.
An enthalpy change describes the change in enthalpy observed in the constituents of a thermodynamic system when undergoing a transformation or chemical reaction. The breaking of bonds between solute molecules the breaking of intermolecular attractions between solvent molecules and the formation of new solute-solvent attractive bonds. First of all the heat change in the calorimeter formula is normally represented by a lowercase Q but it can also be represented by change in enthalpy or delta H because remember at constant pressure delta H equals q and constant pressure is almost always a good assumption for the duration of an experiment or at least as long as we stay at the surface of the earth.
Generally enthalpy of bond dissociation values differ from bond enthalpy values which is the average of some of all the bond dissociation energy in a molecule except in case of diatomic molecules. The idea here is that you can use the heat absorbed by the solution to find the heat given off by the dissolution of the salt. The experiments are easy to carry out in a laboratory and the usual equations.
To find the standard enthalpy change of reaction DeltaH_rxn using the standard enthalpies of formation you need to take into account the fact that for a given chemical reaction the change in enthalpy is independent of the pathway taken. Enthalpy change is the difference between the energy contents of the products and reactants when a reaction occurs. Enthalpy change of solution The enthalpy change of solution is the enthalpy change when 1 mole of an ionic substance dissolves in water to give a solution of infinite dilution.

Lattice Enthalpies And Enthalpies Of Solution
1
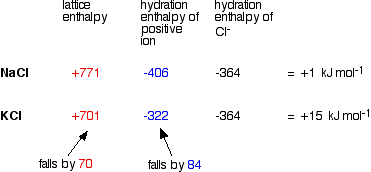
Problems In Explaining The Solubility Of Group 2 Compounds
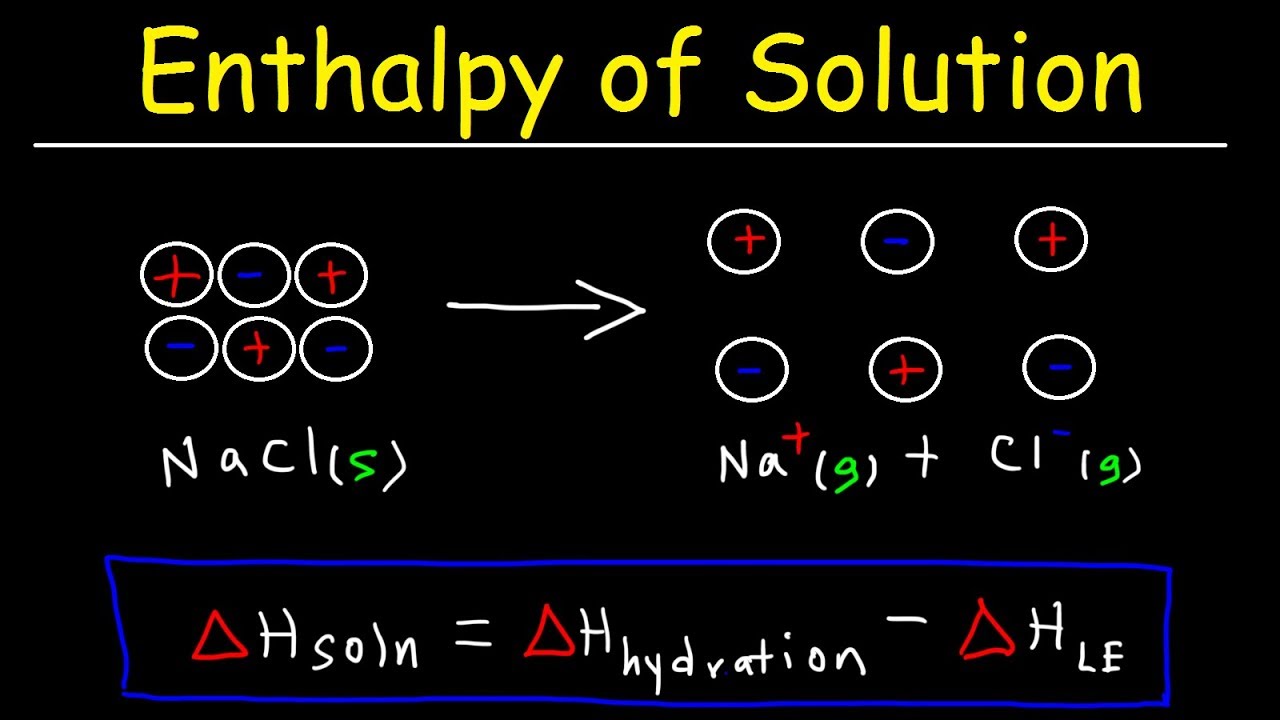
Enthalpy Of Solution Enthalpy Of Hydration Lattice Energy And Heat Of Formation Chemistry Youtube

Enthalpy Of Solution
Energy Changes In Solutions
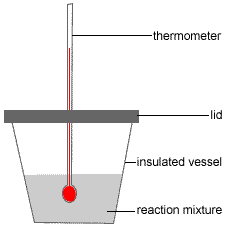
Heat Of Reaction Chemistry Tutorial
How Do I Work Out Standard Enthalpy Change Of Solution The Student Room
