Oleyl alcohol ˈ oʊ l i ˌ ɪ l ˈ oʊ l i əl or cis-9-octadecen-1-ol is an unsaturated fatty alcohol with the molecular formula C 18 H 36 O or the condensed structural formula CH 3 CH 2 7-CHCH-CH 2 8 OH. Haloalkane or alkyl halides are the compounds which have the general formula RX where R is an alkyl or substituted alkyl group and X is a halogen F Cl Br I.
Unit 2 8 Alcohols And Halogenoalkanes Notes

Laboratory Synthesis Of Alcohols From Halogenoalkanes Haloalkanes Reagents Reaction Conditions Equations Advanced A Level Organic Chemistry Revision Notes Doc Brown

Haloalkane An Overview Sciencedirect Topics
Ethanol CH 3 CH 2 OH also called ethyl alcohol is a particularly important alcohol for human use.

Haloalkane to alcohol. Alcohol can be converted to haloalkanes. The chlorofluorocarbon compounds of methane and ethane are collectively known as freons. In a primary alcohol the carbon bonded to the OH group is also bonded to only one other carbonIn a secondary alcohol and tertiary alcohol the carbon is bonded to two or three.
Dipole-dipole force Intermolecular forces found between polar molecules ie. A tertiary halogenoalkane haloalkane or alkyl halide is formed. As fire extinguisher under the name pyrene.
71 Nucleophilic Substitution Reactions of Haloalkanes Nucleophilic substitution reactions are ionic reactions that break and make chemical bonds by transfers of pairs of electrons. I n the alcohol functional group a carbon is single-bonded to an OH group the OH group by itself is referred to as a hydroxylExcept for methanol all alcohols can be classified as primary secondary or tertiary. Ethanol is the alcohol produced by some species of yeast that is found in wine beer and distilled drinks.
It can be produced by the hydrogenation of oleic acid esters by BouveaultBlanc reduction which avoids reduction of the CC. 20 Ab initio calculations were carried out to. Carbon tetrachloride is used.
A 6-endo-tet ring closure of an epoxy alcohol 642 Scheme 616 to a tetrahydropyran 643formally a violation of Baldwins rules of ring closure which predict that a 5-exo-tet ring closure is favored 19 was catalyzed by a catalytic antibody indicating that catalytic antibodies may have utility in catalyzing unfavorable reactions. Dehydration Elimination of water from a compound usually such as an alcohol. A tertiary alcohol reacts if it is shaken with concentrated hydrochloric acid at room temperature.
The OH group by itself is referred to as a hydroxyl. Dehydrohalogenation The elimination of hydrogen and a halogen from a haloalkane. In the alcohol functional group a carbon is single-bonded to an OH group.
It is a colorless oil mainly used in cosmetics. CCI 4 is a colourless non-inflammable poisonous liquid soluble in alcohol and ether. Haloalkanes have been known for centuries.
Specific chemical transformations such as the conversion of a haloalkane R-X into an alcohol R-OH. If you dont know what primary secondary and tertiary alcohols are you should read the introduction to alcohols before you go on. Molecules in which there is an.
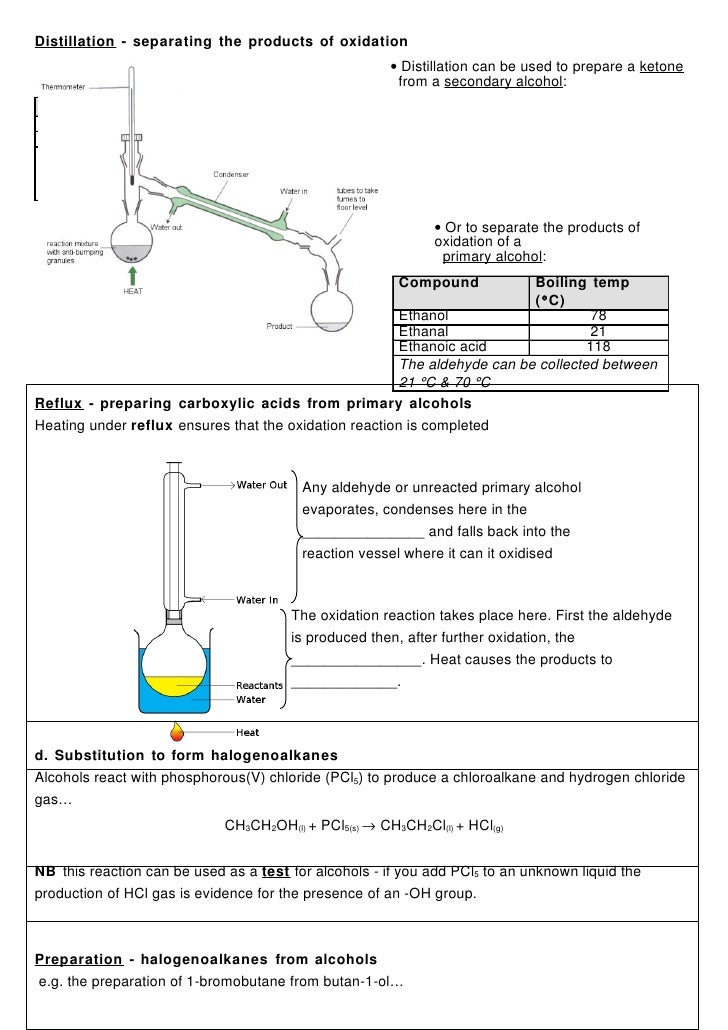
-X Cl Br I or F Haloalkane Alcohol Ether-NH 2 Aldehyde Ketone Carboxylic Acid Ester Amide Amine-ane-ene-yne not responsible halo--ol not responsible-al-one-oic acid-oate-amide amino-Nomenclature Review methan- ethan- propan- butan- pentan- hexan- heptan- octan- nonan- decan-C C C C C C C C C C H O O R OH O O NH 2 OR O 1C 2C 3C 4C 5C 6C 7C. As a solvent for oils fats resins. Alcohol aldehyde ketone carboxylic acid dihalogenoalkane diol Br2 Cl2 room temp EAdd HBr HCl room temp EAdd KOH alcoholic heat under reflux Elimination conc.

Hydrolysis With Water Classification Tertiary Secondary Primary Haloalkanes Halogenoalkanes Reactivity Trends With Silver Nitrate Alkyl Halides Advanced A Level Organic Chemistry Revision Notes
Preparation Of Haloalkanes From Alcohols

15 Haloalkanes Flashcards Quizlet

Every Haloalkane Reaction Flashcards Quizlet
Organic Halogen Compounds

Halogenoalkane Reactions Organic Chemistry

Hydrolysis Halogenoalkanes Hydroxide Ion Sn1 Sn2 Nucleophilic Substitution Reaction Mechanisms To Give Alcohols Reagents Reaction Conditions Organic Synthesis
Learn Preparation Of Haloalkanes From Alcohols In 3 Minutes

